|
|||||||||||||||||||||||||||||||||
|
MANY NEW DYERS are often nervous about Acid dyes when they first hear of them; they think the dyes themselves are harsh flesh eating caustic acids. In reality the acid dyes available to the home dyer are non-caustic and very safe to use. A few are even safe enough to eat - such as those sold as food coloring or used in popular drink mixes (please don't eat the dyes). Most broad use "over the counter" all-purpose dyes, like Rit® and Jacquard's all-purpose iDye, have an acid dye component included to dye protein fibers. When home dyeing, Acid Dyes should, of course, be handled with the normal care that you would handle any concentrated powdered dye or craft chemical, which is wear a dust mask when mixing the powders, wear gloves, use good housekeeping procedures, you know the drill. Acid Dyes are used to dye protein fibers such as silk, wool, angora, alpaca, mohair, feathers, etc., and even man-made Nylon, which is chemically similar to silk. The "acid" part of the name comes into play because a very mild acid like household white vinegar, or odorless Citric Acid, is used to lower the pH of the dye bath so it is slightly acidic, which causes the dye to bond to the protein fibers. This is opposite to using Fiber Reactive dyes on cellulose fabrics, where the dye bath has to be alkaline. You could even say that Acid Dye is better for protein fibers than other types of dye, as proteins respond better to mildly acidic dye baths than to alkali; they tend to stay softer, and silk keeps its sheen. Folks who regularly work with silk often recommend vinegar rinses to restore softness to dyed silk. For you chemistry buffs, Acid Dyes are so called because they contain acidic molecular groups such as -S03H and work in a low pH environment with a mildly acidic "fixative" like white vinegar or citric acid as mentioned above. Acid Dyes are used to dye protein fibers (and nylon) which are all made out of proteins with amino groups -NH2 and the bond between the dye and fiber occurs between the basic amino groups and acidic -S03H groups. Acid dyes are thought to fix to fibers by hydrogen bonding, Van der Waals forces and ionic bonding.
|
|||||||||||||||||||||||||||||||||
|
The structure of the acid dye molecules fall into four main categories: Azo, Anthraquinone, Premetalized and Triphenylmethane. If you don't have a degree in chemistry these names may not mean much to you but they refer to the main molecular groups that make up the dye. It gets a bit more confusing because sometimes dyes fall into more than one of these categories. The main thing to remember is they all have that -S03H that likes to attach to the amino groups within the fiber. |
|||||||||||||||||||||||||||||||||
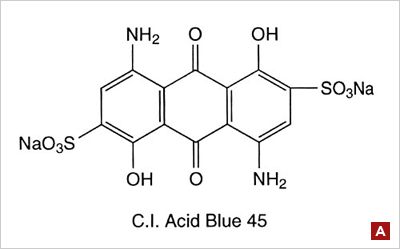 |
Anthraquinone type: Many blue dyes have this structure as their basic shape. The structure predominates in the Leveling class of acid dye. (figure A) | ||||||||||||||||||||||||||||||||
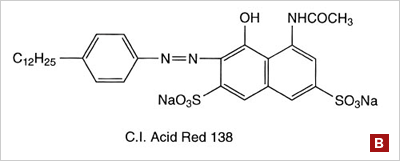 |
Azo dyes: The structure of azo dyes is based on azobenzene, Ph-N=N-Ph. Officially, Azo dyes are a separate class of dyes primarily used in on cellulose fibers such as cotton and rayon, but many acid dyes have a similar structure; most give different shades of red. (figure B) | ||||||||||||||||||||||||||||||||
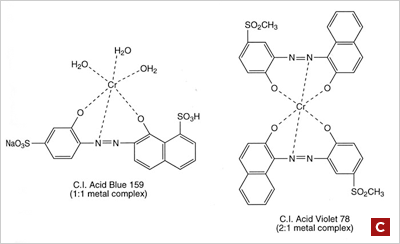 |
Premetallized acid dyes fall into two classes, 1:1 metal-complexes, in which one dye molecule is complexed with one metal atom and the more modern 1:2 metal complexes, in which one metal atom is complexed with two dye molecules. The dye molecule is typically a monoazo structure containing additional groups such as hydroxyl, carboxyl or amino groups, which are capable of forming strong co-ordination complexes with transition metal ions, typically chromium, or cobalt. (figure C) | ||||||||||||||||||||||||||||||||
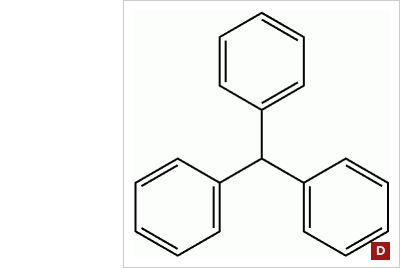 |
Triphenylmethane dyes: Acid dyes having structures related to triphenylmethane predominate in the milling class of dye. There are many yellow and green dyes commercially applied to fibers that are related to triphenylmethane. (figure D) |
||||||||||||||||||||||||||||||||
|
Acid Dyes fall into 3 subgroups or classes that describe their properties in the dyeing process. The three classes are leveling, milling/supermilling and premetallized. Leveling Acid Dyes work best out of all of the acid dye classes for dyeing very even solid colors, and the colors are very bright. The molecules are simple and small and migrate fastest through the dye bath or the fiber, which is why they dye so evenly. They have a few drawbacks, not the least of which is that they are often not as wash fast as the other types of acid dye, so they should be washed in cool water or dry cleaned. (That is good practice with dyed silk and wool anyway!) Products such as our Dharma Dye Fixative or Retayne in a hot soak can improve their washfastness. Even though they sometimes have washfastness levels of 4-5 on a scale of 1-5, they are tested for washfastness at much cooler temperatures than something like Fiber Reactive Procion type dyes, and they will bleed in hot water. These require the most acidic dye bath of the three types, but as you can see from the chart below, if you work from a standard set of directions and your ph runs around 4, all three classes of acid dyes will work. Some folks recommend the addition of Glaubers Salt as a leveling agent with these Leveling Acid Dyes, though this is the only class of acid dyes that Glaubers Salt would work with. It actually does the opposite with Milling and Premetalized Dyes, which work better with Ammonium Sulfate. This leveling class of dyes often doesn't yield the same shades or strength on silk as wools, or even on different types of wool, so mixes can skew on silk vs. wool. Best to never dye different fibers in the same dye pot, as the wool will suck up the dye and leave little for the silk. Milling/Supermilling Acid Dyes are larger molecules with a higher molecular weight then Leveling acid dyes, and they get their name for their excellent wash fastness. They are more washfast than Leveling Acid Dyes. Milling is a process used when felting and fulling wool which requires a lot of hot water and agitation, so dyes used on wool to be felted need to be especially wash fast. Their lightfastness varies. Their main draw back is that they do not dye quite as evenly as Leveling Acid Dyes so extra care must be taken in preparation of the dye bath to ensure even results. Proper agitation of the dye bath and the addition of Ammonium Sulfate (found in your local garden shop as a fertilizer) instead of vinegar can help. This actually "slows" the reaction process, as it starts at a neutral PH, then becomes acidic as the dye bath heats, allowing the larger molecules time to penetrate the fiber more evenly. These are sometimes called "Neutral" Acid Dyes as they don't require as acidic of a dye bath. Premetallized Acid Dyes are even larger acid dye molecules complexed with an ion of a metal, usually chromium or cobalt in a trivalant form, which is relatively safe when handled properly, and present in such minute quantities that by the time it is diluted with water, it more than meets EPA requirements for disposal. These dyes are very wash fast and do not migrate once they fix to the fiber, so you must be extra extra careful to apply them evenly. Proper agitation of the dye bath and the addition of Ammonium Sulphate as a leveling agent, just like with the Milling Acid Dyes, should solve this. These are the most wash fast and light fast of all of the classes of acid dyes. These dyes work especially well on dyeable Nylons and other synthetic poly-amide fibers. These dyes have a particularly high affinity for wool, but in our experiments yielded exactly the same colors on silk, even with mixed colors, which doesn't always happen with the other types of acid dye, especially the Leveling types. They take a great range of ph in the dye bath.
|
|||||||||||||||||||||||||||||||||
|
So what type of acid dyes are Dharma's new dyes? Well, we have brought in some of each. Other lines of acid dye sold for home/light commercial use mix types also, as there is no other way to have a good range of colors. We have several pure colors of Leveling dyes and a couple of Milling dyes that you may or may not find in other lines, plus our own proprietary mixes of those, and we have a bunch of newer 1:2 Premetallized dyes and mixes of same that most of you out there have never had a chance to try. One exception is our #424 True Turquoise, which is actually a Direct dye (Direct Blue or Turquoise 86), but works as well as an acid dye on protein fibers, with the combination of the heat and the acid fixative. It is the most wash fast of all the Direct dyes, and worked very well in our tests on wool and silk, and gives you a wonderful color that simply isn't available in the acid dye line. It is also very light fast. Generally we picked dyes with the best washfastness ratings, as we thought that was the most desirable feature, with a couple of exceptions from the Leveling group that we felt we had to have to give more color choices. We thought we would like to have more choices, so you serious dyers out there can experiment with some types or colors you haven't had access to before. In most cases, our mixes were developed with pure colors from within the same groups, i.e. leveling + leveling, so that they will work well as a mix. But, just as we experience with Fiber Reactive Procion type dyes, even within each type, colors will strike at different rates, migrate through the dye bath or through the fabric at different rates, so that is where the artistic experimentation and discovery come in, and some of the more interesting effects. In general, pick the colors you like, use our basic instructions which can apply to all the colors, and then try different combinations and leveling agents. The moral of the story is play, play, play!!!
|
|||||||||||||||||||||||||||||||||